In 2015 the Nobel Prize in Physiology or Medicine went to Chinese scientist Youyou Tu for her discovery of a novel malaria treatment rooted in traditional Chinese medicine. Tu isolated the drug artemisinin from an herb used to treat malaria in China for more than 2,000 years. Today the World Health Organization recommends artemisinin combination therapy as the first line of defense against malaria cases worldwide.
That same year, half way across the world, two UH engineering professors, Peter Vekilov, John and Rebecca Moores Professor of chemical and biomolecular engineering and chemistry, and Jeffrey Rimer, Ernest J. and Barbara M. Henley Associate Professor of chemical and biomolecular engineering, published a study in the Proceedings of the National Academies of Sciences (PNAS) shedding new light on how some of the most effective malaria drugs – those from the quinoline class – prevent the growth of hematin crystals, a key piece of the infection process, from taking place.
The timing of the UH discovery couldn’t be better. Although the use of artemisinin-based treatments has reduced global malaria deaths from nearly 1 million in 2010 to less than 450,000 in 2015, malaria-causing parasites have been known to adapt quickly to current drug treatments, making the need for rapid drug development a priority among medical researchers.
“We have to find drugs faster than the parasite adapts,” Vekilov said.
To do that, Vekilov and Rimer dug deep into the physiology of the malaria parasites and how antimalarial drugs work to stop hematin crystallization, pointing to new directions for malaria drug development. Now their findings are published, once again, in PNAS.
How the disease takes hold
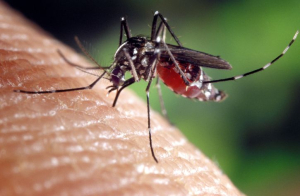
Plasmodium falciparum, one of the parasites that cause malaria, usually infects its hosts through a mosquito bite. Once the parasite enters the host’s red blood cells it begins to consume hemoglobin, producing the toxic byproduct heme.
The plasmodium has evolved to segregate the heme molecules into tiny crystals, about one micron in size. Instead of killing the parasite, the toxic heme molecules attach to sites along the surface of the crystals, allowing the plasmodium to replicate freely inside the host.
Scientists generally understand that antimalarials work by inhibiting the growth of hematin crystals, creating a toxic concentration of heme molecules and killing off the plasmodium.
“But the mechanisms of this crystal inhibition were completely unknown,” said Vekilov.
Until now.
The prevailing theory was that antimalarial drugs prevented crystal growth by binding to the free heme molecules in the solution, but that theory would be proved wrong by Rimer and Vekilov. The pair developed an innovative platform to study hematin crystal growth at the microscopic level in nearly in-situ conditions. Then they showed a significantly higher and possibly toxic concentration of drugs are necessary to inhibit the growth of hematin crystals by binding to the free heme molecules.
Instead they found the drugs bind at six specific sites at the surface of the crystals, blocking the attachment of hematin. Eventually the accumulation of toxic heme in the parasite’s digestive vacuole spells its death.
The duo’s latest paper looks more closely at these binding sites with an eye on designing new, more effective drug treatments for the deadly disease.
“People had speculated that some sites might be better to block than others and we definitively showed that certain modes of inhibition were better than others,” said Rimer. “That might be influential in how we view the design of new antimalarial drugs in the future.”
Co-authors on the paper include former graduate student Katy Olafson, now a postdoc at MIT, and former undergraduate student Tam Nguyen, who is now pursuing his Ph.D. at the UH Cullen College. “They really led the experimental thrusts of the research. They first disproved a popular theory, then proved this new theory. It’s very cool,” said Vekilov.
Staying ahead of the parasite
Despite the success of artemisinin-based therapies in treating malaria, the need for new malaria drugs hasn’t vanished. Before Tu’s discovery of the drug, a drug called Chloroquine was used to treat the disease. Overuse of Chloroquine caused plasmodium parasites to evolve, growing pumps that extracted the drug from their bodies.
Artemisinin-resistant parasite strains have emerged in South East Asia in the last two years. With the use of artemisinin-based therapies on the rise, it’s only a matter of time before the resistant strains spread globally, rendering the treatment ineffective.
“The platform we developed means we can stay ahead of the parasite because it will keep adapting,” said Vekilov.
The researchers see their biomimetic platform for studying hematin crystallization as a new beginning in the accelerated development of antimalarial drugs.
“It’s really an enabling tool,” Rimer said. “Our hope is that we can help guide pharmaceutical companies in selecting molecules that would have the highest probability of success in treating the disease by inhibiting crystal formation.”